
Laura Martínez Muñoz graduated in Biology from the Complutense University of Madrid in 2001 and joined the Hospital Universitario de la Princesa (Madrid) that same year to work on the cloning and characterisation of new genes. In October 2004, she joined Dr. Mario Mellado’s group in the Department of Immunology and Oncology at the National Centre for Biotechnology of CSIC as an FPI fellow. Her thesis project focuses on the study of chemokine receptor conformations. She works on fine-tuning the various resonant energy transfer techniques, demonstrating that the dimeric complexes are tremendously dynamic and that they are regulated, not only by the expression levels of the receptors but by their own ligands. In 2009 she defended her doctoral thesis and began to study the functional relevance of the different conformations described (post-doctoral contracts), obtaining several publications from international collaborations and a co-directed thesis. In parallel, she is developing new techniques (SRET, BRET-BiFC, and TIRFM) that allow her to go a step further in studying the receptor complexes and their function. ComFuturo Researcher (I edition) in the line of research of the programme “Autoimmune inflammatory diseases“. She first developed her project at the National Centre for Biotechnology and then at the Andalusian Molecular Biology and Regenerative Medicine Centre “Chemokine-based therapeutic strategies for the treatment of chronic inflammatory diseases“.

Project Summary
Chemokine receptors form oligomers on the cell surface, dynamic structures that are critical for the function of these mediators and are regulated by receptor levels and the presence of ligands. Of these receptors, CXCR4 and its ligand CXCL12 play a determining role in the homeostasis of the organism and several inflammatory and autoimmune pathologies. CXCL12 is abundantly expressed in the synovial tissues of patients with rheumatoid arthritis (RA), and the incidence of RA observed in murine models of the pathology is much lower in mice with CXCR4-deficient T cells. In this project, we propose to take advantage of the dimerising capacity of these receptors to block or alter their function, thus establishing a proof of concept of a new form of therapeutic intervention to replace the classical one of blocking ligand binding to the receptor which, on the other hand, has proved to be sterile in the clinic. In a study to define new CXCR4 antagonists, we have identified a peptide corresponding to the VI transmembrane domain of CXCR4 (239KTTVILILA). On the other hand, the mutation of 3 residues in this domain of CXCR4, K239E, L244A, L246A gives rise to a non-functional mutant receptor that is able to dimerise with the native CXCR4 and has negative dominance over it. In this project, we aim to achieve the expression of this mutant receptor or the peptide specifically in CD4+ T cells and in the inflammatory focus and evaluate whether this strategy improves the RA symptoms. For this purpose, we will design lentiviral particles that express the HIV-1 envelope protein gp120IIIB and thereby ensure specific infection of CD4+ /CXCR4+ cells while leaving other cell types intact. The introduced genes will be integrated into the genome of these cells and will express the mutant receptor or hydrophobic peptides in their membranes.
Application:
Rheumatoid arthritis is one of the most common autoimmune diseases in developed countries, with a prevalence of 0.3–1.2%. It is characterised by chronic inflammation in the joints as a result of an influx of large numbers of immune system cells into the joints. The aim is to develop new therapeutic tools that, through the specific blocking of cell movement, improve inflammation in patients with this disease.
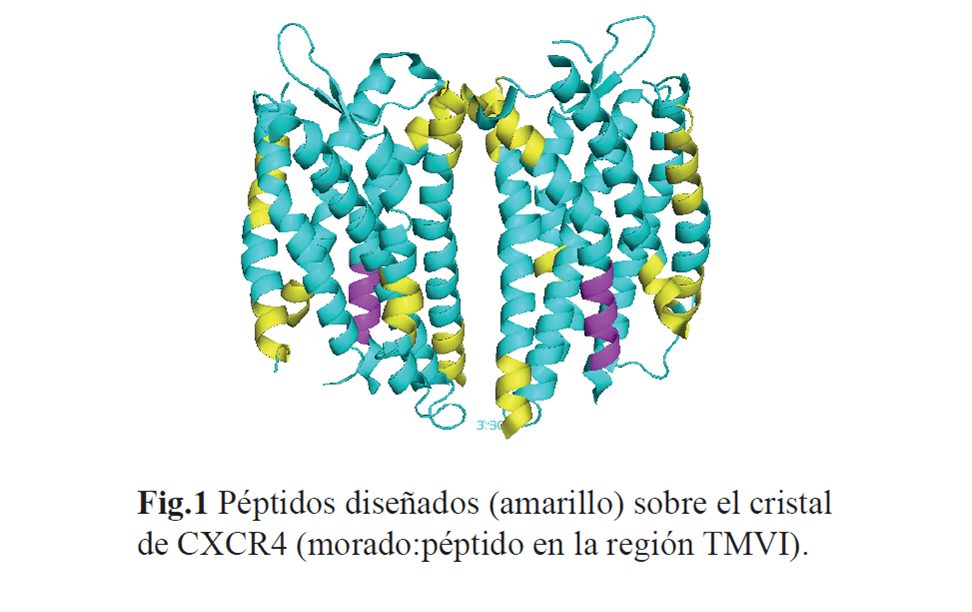
Scientific production derived from the ComFuturo Project
Scientific articles
- L. Martínez-Muñoz; J.M. Rodríguez-Frade; R. Barroso; C.O.S.Sorzano; J.A. Torreño Piña; C.A. Santiago; C. Manzo; P. Lucas; E.M. García Cuesta; E.M. Gutierrez; L. Barrio; J. Vargas; G. Cascio; Y.R. Carrasco; F. Sánchez Madrid; M.F. García Parajo; M. Mellado (2018) Separating actin-dependent chemokine receptor nanoclustering from dimerization indicates a key role for clustering in cxcr4 signaling and function. MOLECULAR CELL. DOI: 10.1016/j.molcel.2018.02.034
- L. Martínez-Muñoz; R. Villares; J.L. Rodríguez-Fernández; J.M. Rodríguez-Frade; M. Mellado (2018) Remodeling our concept of chemokine receptor function: From monomers to oligomers. J LEUKOC BIOL. DOI: 10.1002/JLB.2MR1217-503R
- P.J. Collins; M.L. McCully; L. Martínez-Muñoz; C. Santiago; J. Wheeldon; S. Caucheteux; S. Thelen; V. Cecchinato; J.M. Laufer; V. Purvanov; Y.R. Monneau; H.Lortat-Jacob; D.F. Legles; M. Uguccioni; M. Thelen; V. Piguet; M. Mellado B. Moser (2017). Epithelial chemokine CXCL14 synergizes with CXCL12 via allosteric modulation of CXCR4. THE FASEB JOURNAL. DOI: 10.1096/fj.201700013R
- J.M. Rodríguez-Frade; A. Guedán; P. Lucas; L.Martínez-Muñoz; R. Villares; G. Criado; D. Balomenos; H.T. Reyburn; M. Mellado García (2017). Use of lentiviral particles as a cell membrane-based mFasL delivery system for in vivo treatment of inflammatory arthritis. FRONTIERS IN IMMUNOLOGY. DOI: 10.3389/fimmu.2017.00460
- J.M Rodríguez-Frade; L. Martínez-Muñoz; R. Villares; G. Cascio; P. Lucas; R.P. Gomariz,; M. Mellado (2016). Chemokine Detection Using Receptors Immobilized on an SPR Sensor Surface. METHODS IN ENZYMOLOGY. DOI: 10.1016/bs.mie.2015.09.013
- R.A. Lacalle; J.C. de Karam; L. Martínez-Muñoz; I. Artetxe; R.M. Peregil; J. Sot; A.M. Rojas; F.M. Goñi; M. Mellado; S. Mañes (2015). Type I phosphatidylinositol 4-phosphate 5-Kinase homo- and heterodimerization determines its membrane localization and activity. FASEB JOURNAL: OFFICIAL PUBLICATION OF THE FEDERATION OF AMERICAN SOCIETIES FOR EXPERIMENTAL BIOLOGY. DOI: 10.1096/fj.14-264606
- M. Mellado; L. Martinez-Muñoz; G. Cascio; P. Lucas; J.L. Pablos; J.M. Rodríguez-Frade (2015). T cell migration in rheumatoid arthritis. FRONTIERS IN IMMUNOLOGY. DOI: 10.3389/fimmu.2015.00384
Capítulos de libro
- L. Martínez-Muñoz; J.M. Rodríguez-Frade; M. Mellado (2016). Use of Resonance Energy Transfer Techniques for In Vivo Detection of Chemokine Receptor Oligomerization. “Chemotaxis”, Methods and Protocols. Methods in molecular biology (Clifton, N.J.). 1407. 341 – 400. ISBN/ISSN: 1940-6029
Works presented at conferences
- L. Martínez-Muñoz; J.M. Rodríguez-Frade; R. Barroso; C.O.S.Sorzano; J.A. Torreño Piña; C.A. Santiago; C. Manzo; P. Lucas; E.M. García Cuesta; E.M. Gutierrez; L. Barrio; J. Vargas; G. Cascio; Y.R. Carrasco; F. Sánchez Madrid; M.F. García Parajo; M. Mellado. Separating actin-dependent chemokine receptor nanoclustering from dimerization indicates a key role for clustering in cxcr4 signaling and function. 6th International Iberian Biophysics Congress and X Iberoamerican Congress of Biophysics. Invited oral presentation. Castellón (Spain). 20/06/2018-22/06/2018
- L. Martínez-Muñoz; J.M. Rodríguez-Frade; R. Barroso; C. Manzo; J.A. Torreño Piña; M.F. García Parajo; M. Mellado. Actin cytoskeleton and CD4 control CXCR4 nanoclustering and cell function. European Chemokine and Cell Migration Conference. Poster. Cardiff, United Kingdom. 06/09/2017-09/09/2017
- L. Martínez-Muñoz; J.M. Rodríguez-Frade; R. Barroso; C.O.S. Sorzano; J.A. Torreño-Pina; C.A. Santiago; C.Manzo; P.Lucas; L.Barrio; J.Vargas; G.Cascio; Y.R. Carrasco; F. Sánchez-Madrid; M.F. García-Parajo; M. Mellado. CXCR4 transmembrane region VI governs its actin-dependent dynamic clustering, signaling and cell response. 16th SBE Congress (Sociedad de Biofísica de España). Poster. Seville, Andalusia. 06/06/2017-09/06/2017
- L. Martínez-Muñoz; J.A. Torreno Pina; J.M. Rodríguez-Frade; R. Barroso; C.O.S. Sorzano; P. Lucas; J. Vargas; C. Santiago; Y. Carrasco; F. Sánchez-Madrid; M. García Parajo; M. Mellado García. CXCR4 nanoscale organization and dynamics influence CXCL12-mediated responses. ICREA International Symposium. BioNanoVision of celular architecture: from the nucleus to the cell membrane. Poster. Barcelona, Spain. 25/05/2016 – 27/05/2016
Registered patent applications
- Diana terapéutica en receptores de quimiocinas para la selección de compuestos útiles para el tratamiento de procesos patológicos que implican la señalización de quimiocinas. Application number: PCT/ES2018/070484. See brochure
